It can also help scientists assess the potential for the coronavirus to morph in response to human antibodies. “The work is a step toward understanding what it is about these viruses, and their RBDs in particular, that allows them to acquire this capacity to efficiently infect human cells.” More broadly, the research could shed light on how this family of viruses evolved - and how they jumped into humans, Starr added. The work could also guide efforts to create therapies that target certain parts of the RBD. “Our maps immediately give insight to people who are engineering vaccines,” he said.įor COVID-19 vaccines that rely on the RBD, the maps can identify the most stable forms of the protein that could be mass produced, he said. So a deeper understanding of how these antibodies can bind to the virus’s RBD is critical to building an effective vaccine and other antibody-based therapies, Starr said. But many COVID-19 vaccine candidates aim to coax people’s immune systems to make them by exposing them to the RBD and not the deadly virus that makes it. That’s because the RBD is an important part of what our immune systems “see.” Patients who have recovered from COVID-19 naturally produce protective proteins called neutralizing antibodies that target the RBD. Those resources will hopefully help guide efforts to treat COVID-19, said Starr, an evolutionary biochemist. Sarah Hilton, an evolutionary and computational biologist in the Bloom Lab, the team aggregated that data into heat maps and visualizations of how the mutations affected the form and function of the RBD. To take a wide look at RBD mutations, the researchers used an experimental technique called “ deep mutational scanning.” It essentially is a warp-speed way to create a catalog of every possible mutation and then ask how each mutation affects the virus. That can serve as an early detection system for whether unexpected mutational patterns begin to emerge. Scientists worldwide can now instantly “look up” any mutation that is seen in a viral sample taken from a patient. But the RBD may be able to tolerate a number of mutations.”Īs the pandemic progresses, this “atlas of mutations” can serve as a useful reference, she added. “There’s no reason to believe that going beyond that level will make it more pathogenic or transmissible. “The virus already has a ‘good enough’ ability to bind to ACE2,” said Greaney, a graduate student at the University of Washington. And a tighter bind doesn’t necessarily translate to a more dangerous disease. No mutations that strongly increase ACE2 binding have been spotted in patients with COVID-19, she cautioned. The finding could explain the diversity in this domain that has accumulated over the evolution of these viruses - and shed light on their future evolution. But a surprising number either were tolerated or helped the RBD bind more tightly with ACE2, said Greaney, an evolutionary virologist. In the team’s experiments, most of the mutations to the RBD hindered the virus’s binding ability. Their proofreading is imperfect, and mistakes happen. As viruses spread, they make copies of themselves. “Mutations” might sound ominous, but they are a humdrum part of viral existence, the study authors noted. Tyler Starr and Allison Greaney, measured how virtually every possible mutation to the RBD would affect its ability to stably fold and bind to ACE2. Scientists on Bloom’s research team, led by Dr. This work is also important for understanding the virus’s evolution and, eventually, for understanding whether this virus can evolve to evade immunity.” What do the mutations mean? “So understanding how mutations affect the RBD can help guide the design of vaccines. Jesse Bloom, an evolutionary biologist and the study’s senior author. “Almost all vaccines in development include the receptor-binding domain in order to trigger an immune response against the virus,” said Dr. 11 in the journal Cell, aims to shed light on how the coronavirus evolved to easily infect human cells - and to guide efforts to stop it. Scientists at Fred Hutchinson Cancer Research Center have taken the first comprehensive look at how mutations to the RBD would affect that ability to bind to ACE2. One part of that spike - the receptor-binding domain, or RBD - acts like a lock pick, binding to a molecule called ACE2 on human cells and letting the virus slip inside. We’ve all seen images of the spike protein that sits atop the coronavirus like a crown. The deaths, the economic crisis, the relentlessly rising number of COVID-19 cases - it all started with a tiny, now-infamous bit of protein. Viruses, Vaccines and Infectious Diseases.Institutional Partners & Collaborations. 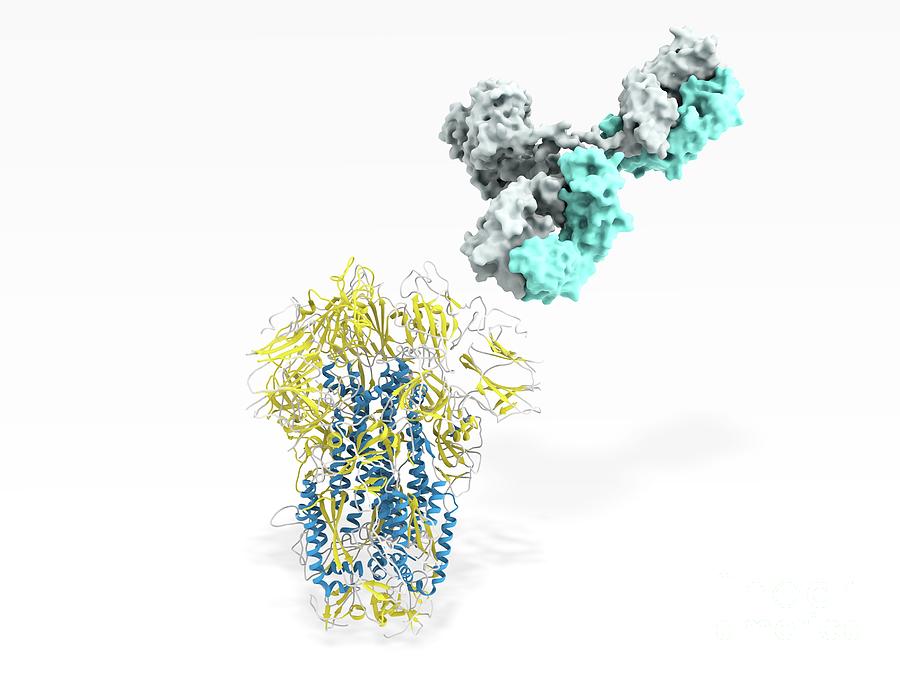
Vaccine and Infectious Disease Division. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
